Time is a large part of geology. The science of geology is fundamentally about the history of the Earth. As it is a historical science, core aspects of geology include chronologies, determining sequences of events, and identifying specifically when significant events occurred.
As for human history, the study of geology would be nearly impossible without ways to tell time. The difference is that time frames involved in geologic history are much, much longer than human timescales.
 |
| An example of how radiometric dates constrain the age of a sedimentary rock layer. Ash beds found near the bottom and top of the Morrison Formation indicate that it was deposited between about 157 and 150 million years ago. |
Early in the development of the geological sciences, researchers determined that Earth must be very old based on observations of geologic processes such as erosion by rivers and deposition of sand on beaches as well as the vastness of the rock record. In the 18th century, Earth’s history was described as having “no vestige of a beginning, no prospect of an end” by James Hutton, the “father of modern geology.” Although early geologists had no way to identify when (e.g., numeric ages) geologic events occurred, they were able to decipher the order of geologic events (relative ages) using principles such as sequential layering of rock layers superposition), fossil correlation, and cross-cutting relationships.
Geologists realized that naturally occurring radioactive elements present in the Earth could be clocks in rocks soon after the discovery of constant rates for radioactive decay. Radioactive decay rates are expressed as half-lives, the time that it takes for half of the radioactive parent isotope to decay into a daughter isotope. The half-life is unique for each radioisotope and radioactive decay rates are not impacted by variables like chemical reactions or temperature.
Radioactive isotopes of several elements including uranium (U), thorium (Th), and potassium (K) serve as timekeepers in geology. These dating techniques are quite complicated in detail, but relatively simple in principle. The least complicated radiometric techniques commonly used to obtain numeric ages of rocks involve a radioactive isotope of potassium (potassium-40). Uranium and thorium dating techniques are more complicated, in part because these elements do not decay in a single step, but instead have complicated decay series that include several short-lived radioisotopes (including radon and radium) that in turn decay before the stable daughter (lead) is formed.
Most potassium is stable, but a tiny percentage (0.012%) is radioactive with a half-life of 1.25 billion years, providing a clock with a pendulum that can measure all but the most recent geologic events. Potassium-40 is also useful to geologists is that it can decay to argon (Ar), a noble gas (like helium) that does not form chemical bonds with other elements. Therefore, when a mineral crystal forms, say in a body of magma prior to a volcanic eruption, it does not contain argon. But through time, radioactive decay of potassium-40 produces argon which is then trapped inside crystal grains.
 |
| A volcanic ash layer in the Morrison Formation. Photo courtesy of Jim Kirkland, Utah State Paleontologist. |
The potassium-argon dating technique involves measurement of the quantity of potassium-40 and argon and calculations using potassium-40’s half-life.
In recent decades, geologists have developed a more advanced version of this technique wherein samples are placed in a research nuclear reactor to convert regular potassium (potassium-39) into argon-39. Argon dating is both more accurate and more precise, allowing geologists to measure the quantity of just one element (argon) and the relative amounts of the different isotopes. Argon dating is one of the most widely used geologic dating techniques in current research.
Like for any scientific analysis, there are constraints on when argon dating can be used. Argon dating cannot be used to date extremely young rocks, because with its long half-life, enough time may not have passed for measurable amounts of argon to form. It also can only be used on minerals (like feldspars) that contain potassium.
 |
| The US Geological Survey’s argon dating laboratory in Menlo Park, California. USGS image. |
Argon dating is performed almost exclusively on volcanic rocks since the time of volcanic eruption is what starts the argon clock. But it can be used on sedimentary rocks that contain volcanic ash layers. Volcanic ash (which is finely shattered rock material produced during eruptions) from distant volcanic eruptions can fall onto river floodplains and other ancient environments and provide timekeepers for sediments above and below them.
Argon dating and other techniques have been used to date volcanic ashes in several rock layers (formations to geologists) near Moab, including the Chinle, Morrison, and Cedar Mountain formations. The ages of rock layers above and below the units containing dated volcanic ashes are inferred based on these available age determinations. Other dating techniques have been used to identify when the La Sal Mountains formed and to determine the age of the Colorado River canyons near Moab.
Together, these clocks in rocks have helped build the timeline of Moab’s geologic history. The oldest rocks found nearby are 1,700 million year old igneous and metamorphic rocks in Westwater Canyon. The Chinle Formation was deposited between 230 and 201 million years ago, and the Morrison Formation was between 157 and 150 million years ago. The current canyon country landscape formed much more recently, within the last 1.5 million years.
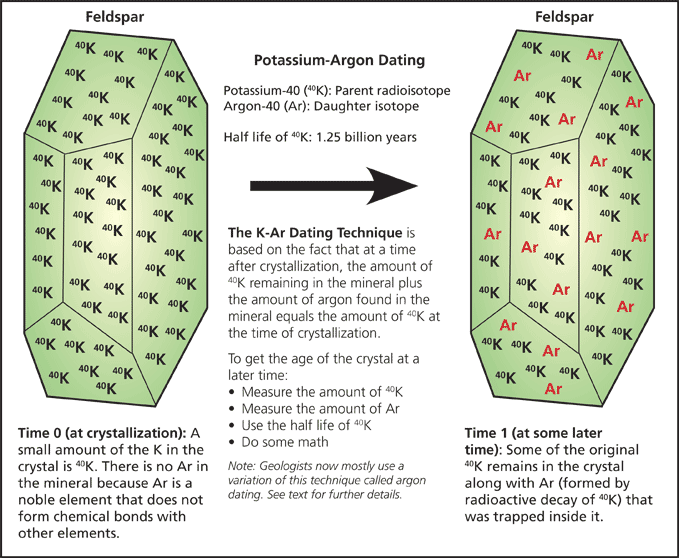 |
| The potassium-argon dating is a relatively straight-forward example of how radiometric dating techniques work. |
|